Why doesn’t the government know what’s in your food? Because industry can declare on their own that added ingredients are safe. It’s all thanks to a loophole in a 57-year-old law that allows food manufacturers to circumvent the approval process by regulators. This means companies can add substances to their food without ever consulting the Food and Drug Administration about potential health risks. Animation by: Phillip Allen. Produced by: Eleanor Bell, Jared Bennett, Erin Quinn & Chris Young.
Video transcript:
In 2008, when an Australian food manufacturer wanted the US federal government’s stamp of approval on the company’s new ingredients, regulators said no. But go inside many American supermarkets and you’ll find products containing them on the shelves.
So how do new ingredients get from the lab to your dinner table?
When companies create new food additives – to improve their product’s texture, taste, appearance, or to extend their shelf life – they have two choices:
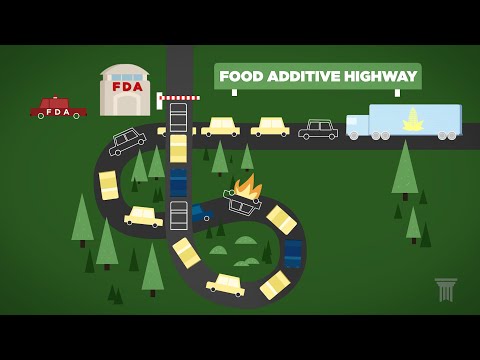
The “Food Additive Highway” is a gridlocked route marked by government potholes. Traffic here is policed by the U.S. Food and Drug Administration – the federal agency that regulates 80 percent of the nation’s food supply.
Companies traveling this path must submit their food additives to extensive review. Then the FDA may issue its formal approval. This journey can take years — even decades — to complete.
So it’s no surprise that companies often take an alternative route.
This road is paved by a legal loophole that hinges on what counts as a “food additive.”
Changes to the law in the fifties created this two-lane system where anything “Generally Recognized As Safe”, or GRAS, travels down a much smoother road to market.
These “GRAS” ingredients are not considered food additives and effectively get a pass to the fast lane.
This “GRAS” clause means companies can determine on their own that what they’re adding to our food is safe.
Then it’s up to the company to inform the FDA if they want to. That’s right. Companies have no legal obligation to tell the FDA what they’re putting in our food.
But if they do decide to pull in for inspection they could get the FDA ‘OK’, which makes them more attractive to potential distributors.
But what if the FDA doesn’t like what it sees?
Take lupin – it’s a legume from same family as peanuts. It’s often used in Mediterranean cooking. It can also be ground into flour and used in gluten free food.
In 2008, when George Weston Foods told the FDA that they’d certified the use of lupin based flour, protein, and fibre in food as safe, regulators at the FDA disagreed. It found that people with peanut allergies could suffer “life-threatening reactions” to lupin-ingredients. FDA officials said ingredient labels listing lupin wouldn’t be enough to protect consumers. The regulators refused to agree that the ingredients were Generally Recognized As Safe.
So George Weston Foods withdrew the notification. But other companies skipped the FDA checkpoint altogether.
The Center for Public Integrity found products containing lupin on supermarket shelves. None included warnings for people who suffer from peanut allergies.
Companies have added at least 1000 ingredients to the food we eat without ever telling the FDA.
Video credits
Production: Eleanor Bell and Jared Bennett
Reporting: Erin Quinn and Chris Young
Animation: Phillip Allen
Voiceover: John Ketchum
Executive Producer: Kimberley Porteous